Knowing the age (i.e. proportion of young vs. old) of eels in an eel population provides an understanding of how fast they grow and may give an indication of the pressures faced by tuna in a particular environment or habitat - pressures such as over-fishing, barriers to migration, density and food quality.
The size of an eel is not always a good indicator of its age, as growth rates tend to be highly variable depending on where they are living and what they are eating. For example, a 500 mm long longfin might be anything from 2 years old - if grown in an area where there are few eels, like a new hydro lake or in culture conditions - to 30 years old, in a cold lake. Large longfin eels are often 30 to 40 years old, but can be more than 90 years in extreme cases.
Once the population size structure - the number/proportion of eels in each size class, such as length - of a selected catchment is known, it is possible to use length-stratified selections of otoliths to estimate the age structure of the population, for example by ageing 10 otoliths in each 10 cm length group per species. Unfortunately, the most accurate ageing and sexing techniques currently available are destructive processes that require killing the tuna. If this is acceptable, the first step is to record the date, site location, species, length and weight measurements of the specimen.
Ageing eels
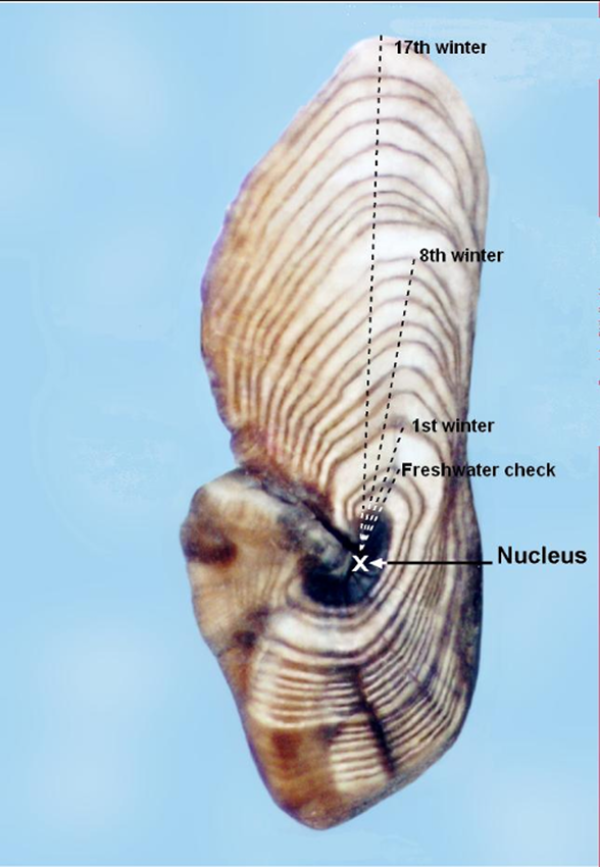
Fish can be aged using measurements of their scales, bones, or other structures such as fin-rays. Currently, the most reliable method to estimate the age of tuna is by examination of their otoliths or "ear bones": small, oval bones found in the head of a tuna. Bones, such as the otolith, grow as the fish grows, with more material being added to the outside of the bone over time as a series of layers.
Growth is much slower during colder months, so that winter growth layers are denser and darker. Currently, the age of tuna is estimated by counting the annual winter growth layers, very much like estimating the age of a tree by counting annual growth rings in a cross section of the trunk.
The results of previous studies using tagged or dye-marked eels, have shown that using otoliths to estimate age is fairly accurate. However, it must be noted that aging is a subjective science and is highly dependent on the experience of the person doing the reading.
Consequently there are on-going discussions regarding the accuracy of the technique in use and its applicability in some habitats.
In addition to estimating age, examination of the otolith may also give an indication of an eel's life history, for instance:
- There is normally a noticeable change or "check" in the centre of the otolith corresponding to when the juvenile (glass eel) moved from the ocean into fresh water.
- Changes in the rate of growth may also be noticeable in the otolith, so that periods of rapid growth are evident when the winter "checks" are widely separated.
The use of otoliths in scientific research is continually evolving and has become very sophisticated.
Microscopic examination of specially prepared otoliths from glass eels has shown that growth rings are deposited daily, so that glass eels can be aged in days. Different parts (e.g. "checks") of an otolith may be separated and analysed for chemical composition. The composition of the material deposited as the otolith grows changes with the chemical composition of the water where the eel lives.
This sort of research can identify when a fish was living in fresh or salt water, and is continuing to develop as the technology becomes available.
There are disadvantages in using otoliths to age tuna:
- It requires killing the fish
- Finding and removing otoliths - it takes practise to develop appropriate skill
- Otoliths are easily lost or damaged, and their preparation and reading is quite involved and time-consuming.
However, if you are going to take an eel to eat, why not remove the otoliths to start gathering the data you may need to learn more about your local tuna populations? You can start collecting them now and send to the laboratory later for analysis.
References and further reading
Arai, T., Otake, T., Jellyman, D.J., Tsukamoto, K. (1999). Differences in the early life history of the Australasian shortfinned eel Anguilla australis from Australia and New Zealand, as revealed by otolith microstructure and microchemistry. Marine Biology 135: 381-389.
Graynoth, E. (1999). Improved otolith preparation, aging and back-calculation techniques for New Zealand freshwater eels. Fisheries Research 42: 137-146. http://www.sciencedirect.com/science/article/pii/S0165783699000296
Jellyman, D.J. (1995). Longevity of longfinned eels Anguilla dieffenbachii in a New Zealand high country lake. Ecology of Freshwater Fish 4: 106-112. http://onlinelibrary.wiley.com/doi/10.1111/j.1600-0633.1995.tb00123.x/abstract