On this page:
- The Earth’s atmosphere
- Greenhouse gases and the greenhouse effect
- Natural and manmade greenhouse gases
- Carbon emissions from burning fossil fuels and agriculture
- Carbon sinks – taking CO2 back out of the atmosphere
- Different greenhouse gases have different effects on the Earth’s atmosphere
- Feedback loops feedback and tipping points
- Common questions and answers about climate change science
The Earth's atmosphere
The atmosphere is the layer of gases (known together as air) that surrounds the Earth. It is kept in place by gravity.
The atmosphere is divided into layers based on temperature and defined by distance from the earth. See more about the layers of the atmosphere.
It is made up of about 78% nitrogen, 21% oxygen, 0.9% argon and 0.04% (400 parts per million or 400 ppm) carbon dioxide (CO2), as well as other gases in tiny amounts (called trace gases). As well as gases, the Earth's atmosphere contains water vapour and aerosols (these are small solid particles or liquid droplets suspended in air).
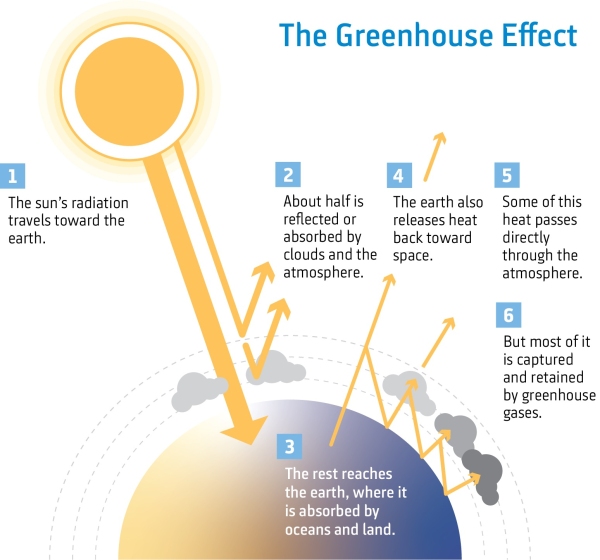
The Earth's atmosphere helps protect all living things from harmful radiation from the sun and space. It keeps the Earth warm by trapping a certain amount of heat. If there was no atmosphere, most of the sun's heat would be reflected back into space and much of the Earth would be frozen. This is known as the greenhouse effect.
Greenhouse gases and the greenhouse effect
A greenhouse gas is a gas in the atmosphere that absorbs heat energy (known as infrared radiation).
Greenhouse gases are important for the Earth's climate because they allow incoming energy from the sun through. However, that energy is transformed into heat at the Earth’s surface and radiates back into the atmosphere. Greenhouse gases absorb some of the outgoing energy and and emit it in all different directions including back to Earth, keeping the surface of the Earth (and the lower atmosphere) warmer than it would be otherwise.
If there were no greenhouse gases in the atmosphere, the average temperature of the earth would be far lower (around -18 degrees Celsius) and much of the Earth would be frozen.
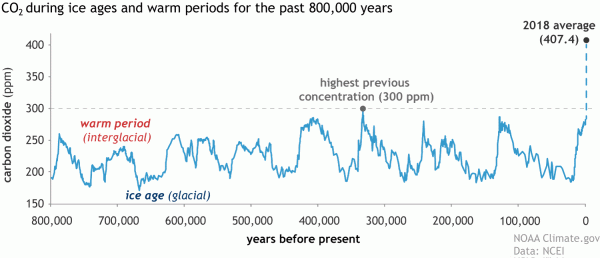
Historically, CO2 has gone down with ice ages and up during interglacial periods. More recently, greenhouse gas concentrations in the atmosphere started rising with the industrial revolution. In 1950 the concentration went above 300 ppm—the highest in at least three million years.
We know about past CO2 concentrations in the atmosphere from ice cores—samples drilled through the ice in places like Greenland and Antarctica. This page on the Science Learning Hub explains how ice cores give us vital information about the past atmosphere.
As you can see in this graph from the US National Oceanic and Atmospheric Administration, current CO2 levels are higher than they have been in at least 400,000 years of Earth's history, and still rising.
Since the industrial revolution began in the late 1700s, humans have emitted more and more greenhouse gases into the atmosphere from things like burning coal and oil for transport and industry, cutting down forests and an increase in land used for agriculture globally. These additional greenhouse gases absorb more radiation, causing the Earth to warm further. This is the underlying cause of our warming global climate.
Natural and manmade greenhouse gases
Some greenhouse gases including CO2, methane, water vapour and nitrous oxide are found naturally in the environment.
But human activities such as burning fossil fuels for industry or transport, and methane from farm animals, are responsible for producing greater quantities of greenhouse gases than occur naturally. Man-made greenhouse gases also include compounds that contain chlorine and bromine such as sulphur hexafluoride and chlorofluorocarbons (CFCs).
Greenhouse gases produced by human activities are known as anthropogenic greenhouse gases.
Carbon emissions from burning fossil fuels and agriculture
Greenhouse gas emissions are often also called carbon emissions. That's because the two most important greenhouse gases—CO2 and methane (CH4)—contain carbon (C).
Both CO2 and methane are produced through the carbon cycle. Human-produced CO2 is primarily produced by burning fossil fuels—oil, gas and coal—taking carbon from deep in the earth and releasing it into the atmosphere.
New Zealand's methane emissions come mostly from sheep and cattle burping – in fact, this makes up around a third of our total greenhouse gas emissions.
You might like to watch the Farms, forests and fossil fuels video from the Parliamentary Commissioner for the Environment, which explores how policy decisions could affect New Zealand's landscapes.
Carbon sinks—taking CO2 back out of the atmosphere
A carbon sink is something that takes CO2 back out of the atmosphere, reducing the overall concentrations remaining in the atmosphere.
Forest sinks
Plants take up CO2 for photosynthesis, enabling them to grow. That makes New Zealand's large native and introduced forests important pieces in our greenhouse gas puzzle.
NIWA has a big research project underway called CarbonWatch NZ. This project is measuring greenhouse gases in the atmosphere at different locations in New Zealand around native and exotic forests, farmland and cities. The goal is to produce a detailed picture of emissions and uptake across different landscapes and inform government policy aimed at reducing our overall emissions.
You can see statistics about New Zealand's annual greenhouse gas emissions and uptake by our forests.
Ocean sinks
Our oceans act as carbon sinks too, and in fact are estimated to take up 30-40% of CO2 produced by human activities.
This extra uptake of CO2 by the oceans is causing ocean acidification, having impacts on marine animals that grow carbonate shells, coral reefs, and growth rates of some fish. Read more about the interactions between the oceans and the climate here.
Different greenhouse gases have different effects on the Earth's atmosphere
Greenhouse gases remain in the atmosphere for different amounts of time. For instance, CO2 is a long-lived greenhouse gas that can remain in the atmosphere for centuries, while methane is a short-lived gas that breaks down within about 9 years. However, methane can absorb much more energy, so it causes much more warming in the short term. The balance of the time in the atmosphere and capacity for energy absorption is described as the 'global warming potential' of a greenhouse gas. For methane, it is around 30 times greater over 100 years than for CO2.
Feedback loops and tipping points
A lot of the uncertainty around the future effects of climate depends on 'feedback loops' and 'tipping points'.
Feedback loops are climate change processes that themselves cause further warming or other impacts. For instance, wildfires can be more severe in a warmer, drier climate, and extensive fires release even more CO2 into the atmosphere, causing further warming.
Tipping points are thresholds in the climate system that, once reached, set off further large changes. For example, scientists think that certain amounts of warming may lead to runaway loss of ice sheets or forests.
Cascading tipping points are where reaching one tipping point causes another, such as thawing of permafrost—permanently frozen ground near the poles and at high altitudes—which might in turn release trapped methane, starting a cascade effect by further raising greenhouse gas emissions and contributing to more warming.
Common questions and answers about climate change science
-

What’s the difference between weather and climate?
While weather and climate are closely related, they’re not the same thing. -

What do 'carbon neutral' and 'carbon footprint' mean?
A carbon neutral activity is one that has a carbon footprint of zero. -

How can carbon emissions be weighed?
The mass of carbon emissions can easily be calculated. -

What are ppm and ppb?
PPM and PPB are units used in atmospheric chemistry to describe the concentration of gases. -

How do we determine past climate?
Information about past climate is obtained from piecing evidence together from various sources. -

Has the climate changed in the past?
The Earth's climate has exhibited marked "natural" climate changes, with time scales varying from many millions of years down to a few years. -

What are Global Warming Potential™ and CO2 equivalent emissions™?
The Global Warming Potential (GWP) of a greenhouse gas is its ability to trap extra heat in the atmosphere over time relative to carbon dioxide (CO2). This is most often calculated over 100 years, and is known as the 100 year GWP. -

El Niño and La Niña
El Niño and La Niña are opposite phases of a naturally occurring global climate cycle known as the El Niño Southern Oscillation, or ENSO for short.